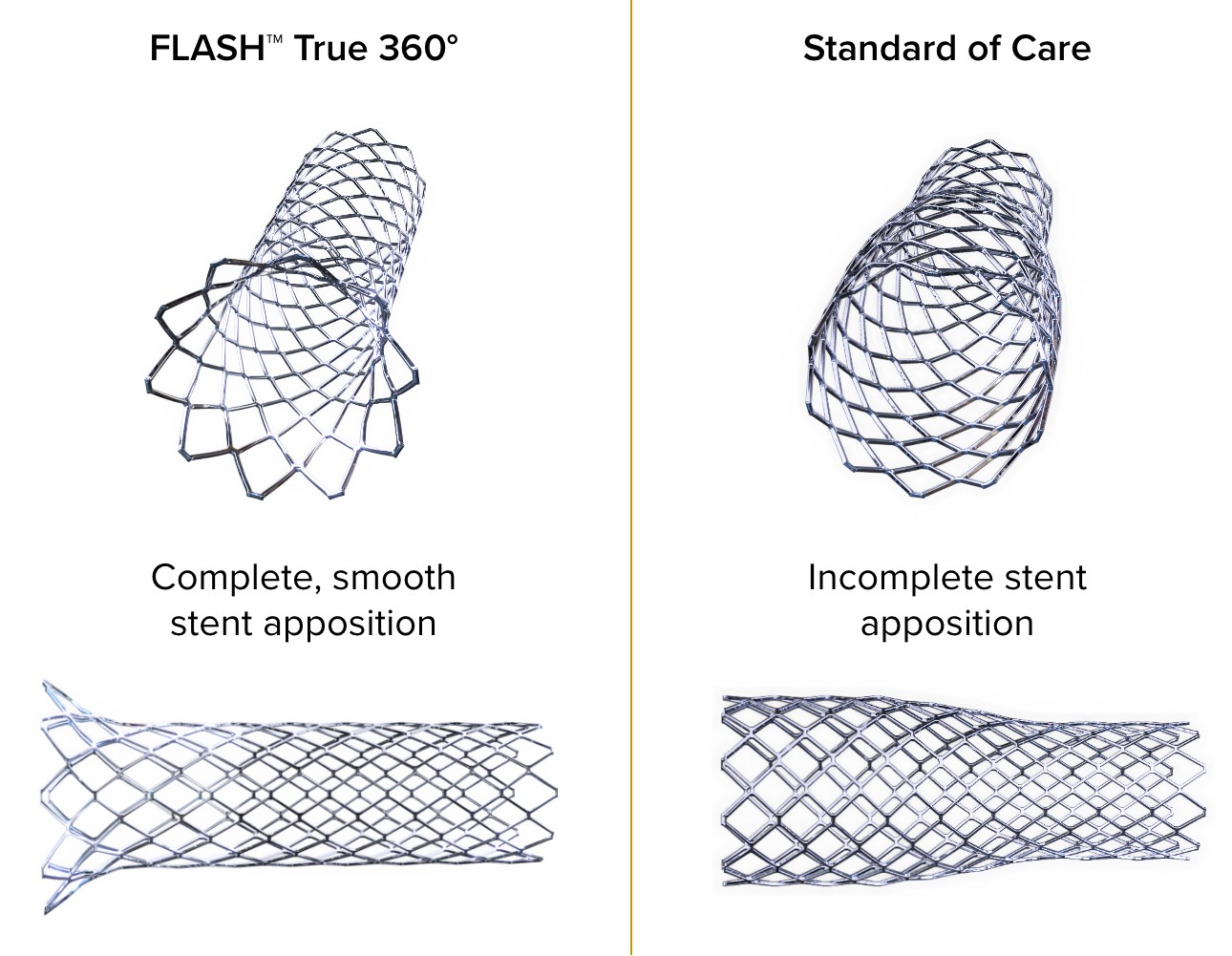
Only FLASH™ achieves total aorto-ostial stent apposition
- Optimize Patient Outcomes
- Total & Efficient Lesion Coverage
- Streamlined Vessel Reaccess
The Challenge
Suboptimal Lesion Coverage
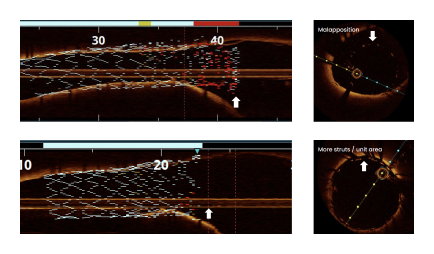
Standard PTCA catheters are unable to create a smooth transition at the ostium creating suboptimal lesion coverage, which is associated with poor outcomes.1
The solution
97%
of lesions demonstrated complete stent apposition2,3
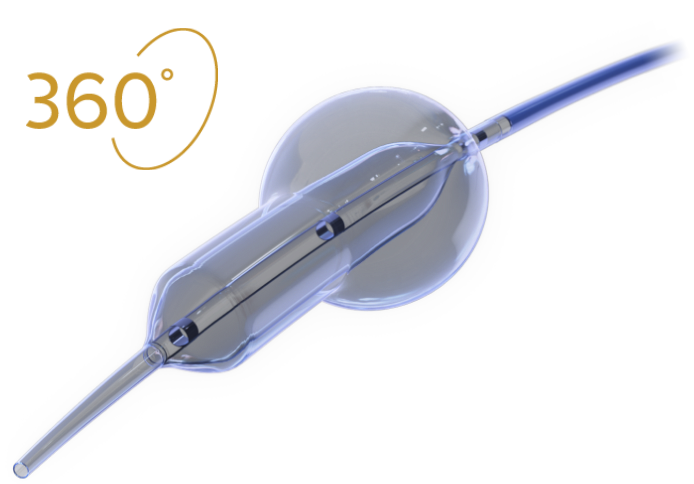
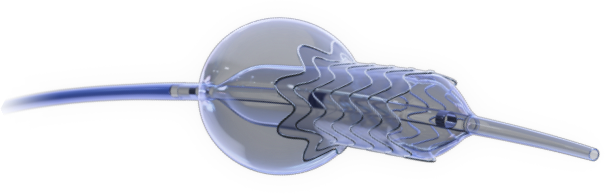
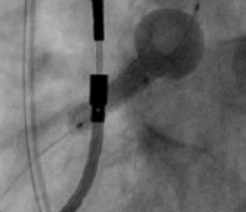
Novel Dual-Balloon Technology
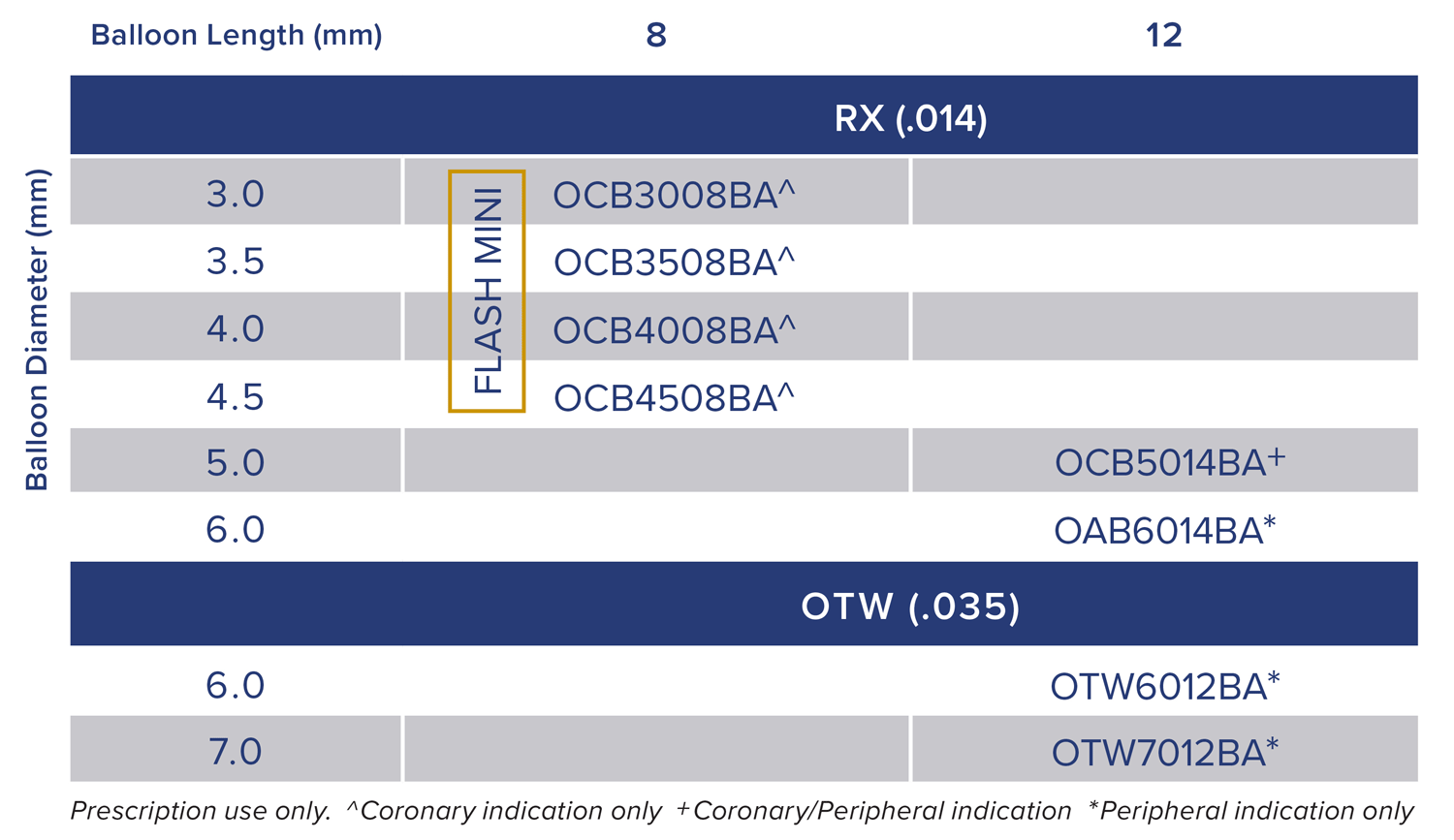
Available for both coronary and peripheral indications.
EASY DELIVERY AND CATHETER POSITIONING
The FLASH system is designed to confidently access and address challenging aorto-ostial stenting, providing total lesion coverage.
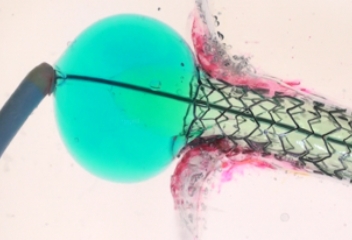
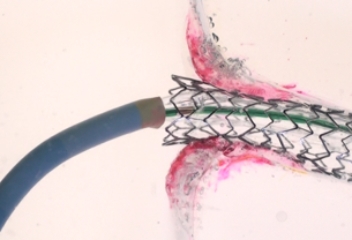
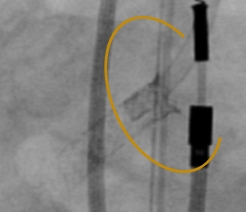
TRUE 360° FLARED APPOSITION
Novel 2-in-1 dual balloon design allows for inflation of a non-compliant distal balloon to anchor the system, while inflation of the compliant, low-pressure proximal balloon conforms the stent to the wall of the ostium.
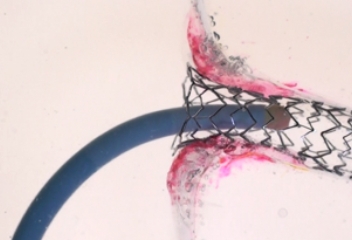
STREAMLINED
REACCESS
With TRUE 360° wall apposition achieved, a smooth transition is created for improved reaccess in future interventions.1,2
confident outcomes
The FLASH System demonstrates consistently low restenosis at 6 months, below the 9% rate reported for accurate stent placement in ostial lesions.6§
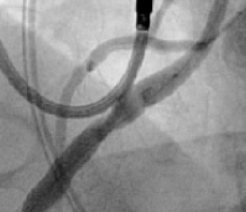
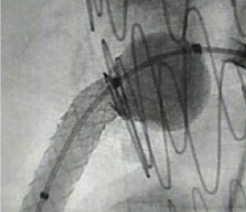
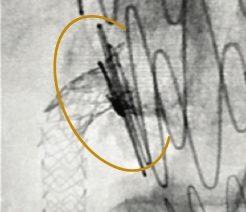
Excellent Final Result
RCA Ostial Stenosis

FEVAR
Resources
Case studies, training videos, and more.
newsroom
Proceeds to accelerate commercialization and pipeline expansion of novel FLASH™ Aorto-Ostial Angioplasty System
Ostial Corporation (Ostial), a private medical technology company focused on addressing the clinical challenges of aorto-ostial interventions, today announced the raise of $7.5M in growth funding led by Delos Capital with participation from AMED Ventures and other existing investors.
Ostial Corporation (Ostial), a private medical technology company focused on addressing the clinical challenges of aorto-ostial interventions, today reached a new milestone, passing the mark of 15,000 commercial units sold of its FLASH™ Aorto-Ostial Angioplasty System (FLASH System) in over 300 hospitals across the United States. FLASH is the first and only dual balloon-based system designed for use in aorto-ostial angioplasty in coronary and peripheral cases.
Read MoreFind out more about FLASH™
§compared to non-ostial lesions
- Cook, et al. Impact of incomplete stent apposition on long-term clinical outcome after drug-eluting stent implantation European Heart Journal 2012. 33:1334-1343.
- Data on file at Ostial Corporation.
- Sanghvi K, Morris T, Kovach R. Optimal Aorto-ostial Lesion Treatment With Flash Ostial Balloon Is Feasible and Safe. JACC. 2016;9(4). Supplement S.
- Nguyen-Trong P, Martinez Parachini J, Resendes E, et al. Procedural Outcomes With Use of the Flash Ostial System in Aorto-Coronary Ostial Lesions. Catheter Cardiovasc Interv.
2016;88(7):1067-1074. - Data on file at Ostial Corporation.
- Dishmon D, Elhaddi A, Packard K, et al. High Incidence of Inaccurate Stent Placement in the Treatment of Coronary Aorto-Ostial Disease. JIC. 2011;23(8).
Indications, Contraindications, Warnings & Precautions
The FLASH™ Ostial system in a specialty balloon device and should only be used by physicians who are trained to use it.
To request a complete copy of the IFU or user training, please contact customer service at customerservice@ostialflash.com.